|
Each element has a symbol, which is one or two letters.The periodic table lists the elements in order of increasing atomic number. 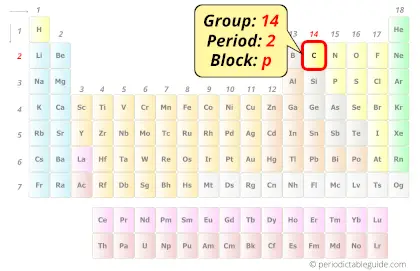
Each element is identified by the number of protons in its atoms.There are 118 elements on the periodic table.The knowledge of the physical forms and properties of non-metals gained in 11–14 chemistry provides a firm foundation for this. They will also learn about chemical reactions involving non-metal elements as reactants and products, such as the Haber process for the production of ammonia, as well as gaining an insight into organic chemistry. Later in their studies, students will learn about the bonding models which give non-metal elements and compounds their diverse properties. This example can help students see that multiple properties of substances need to be used when classifying the substances. But when it is burned, it forms an acidic gas – carbon dioxide – and it has a relatively low density, properties which identify it as a non-metal. A lump of graphite appears shiny, has an extremely high melting point (in fact it sublimes at 3600☌) and will conduct electricity, so on first appearances may be mistaken for a metal. Showing students samples of non-metals which may be mistaken for metals is a useful way of challenging this. They may also use a single property to classify an element, instead of taking an overview of a range of information. However, it is common for students to take the dividing line in the periodic table as absolute and consider metals and non-metals as completely opposite in their properties. There are relatively few misconceptions associated with this topic. Limestone reacts with a dilute acid to generate CO 2 Common misconceptions ***Often described as milky/cloudy, but this loose terminology is often not credited at later assessment stages. **A glowing splint is a splint which is lit, allowed to burn and then blown out leaving a glowing ember at the tip. *Risk assessments must be carried out prior to attempting these procedures with a class – please consult CLEAPSS or similar. Hydrogen peroxide and manganese dioxide catalystīubble the gas through limewater, or pour limewater through a gas sample Suggested simple chemical reaction to generate the gas* If you want to develop your students’ problem-solving skills, use an investigation (eg rsc.li/2UQ8vS9). The gas tests themselves are relatively straightforward and do not take a long time to carry out as a class practical. The most common tests in 11–14 chemistry are for hydrogen, oxygen and carbon dioxide, with tests for chlorine and ammonia featuring in the 14–16 curriculum. Many non-metal elements are gases and there are a number of non-metal compound gases. Gas tests are a key piece of knowledge in this topic. Try to link the narrative about the elements to students’ own experiences, for example graphite used in pencils and the use of chlorine gas in chemical warfare in the first world war. Students are better able to describe and interpret observations when they have a strong knowledge of the common physical forms of substances. Beforehand prepare samples in sealed jars and present them at the start of the topic together with interesting facts about each element. 
Use an element showcase to demonstrate how varied the physical forms of non-metals are compared to metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
